 
The entropy of a system is a measure of its disorder. Standard Enthalpy Change of Combustion is the enthalpy change that occurs when 1 mole of a substance is completely burned in oxygen. ΔH f ° =enthalpy change when 1 mole of a substance is formed from its elements in their standard states Standard Enthalpy Change of Combustion The standard enthalpy change of formation (ΔH f °) is the enthalpy change that occurs when 1 mole of a substance is formed from its elements in their standard states. The standard enthalpy change for the formation of a gas is zero, because the enthalpy of a gas is determined only by its temperature and pressure. This value is usually given in kilojoules per mole (kJ/mol) or in kilocalories per mole (kcal/mol). Standard enthalpy change (enthalpy of formation) is the enthalpy change that accompanies the formation of one mole of a substance from its constituent elements in their standard states. Enthalpy is a measure of the internal energy of a system and is determined by the temperature and pressure of the system. It is calculated by subtracting the enthalpy of the products from the enthalpy of the reactants. The change in enthalpy is a measure of the heat energy transferred in a chemical reaction. The enthalpy of a system is the sum of the internal energy of the system and the product of the pressure and the volume of the system. The enthalpy of a system is the heat content of the system. This law is due to the fact that entropy is a measure of the disorder of a system, and disorder always increases over time. 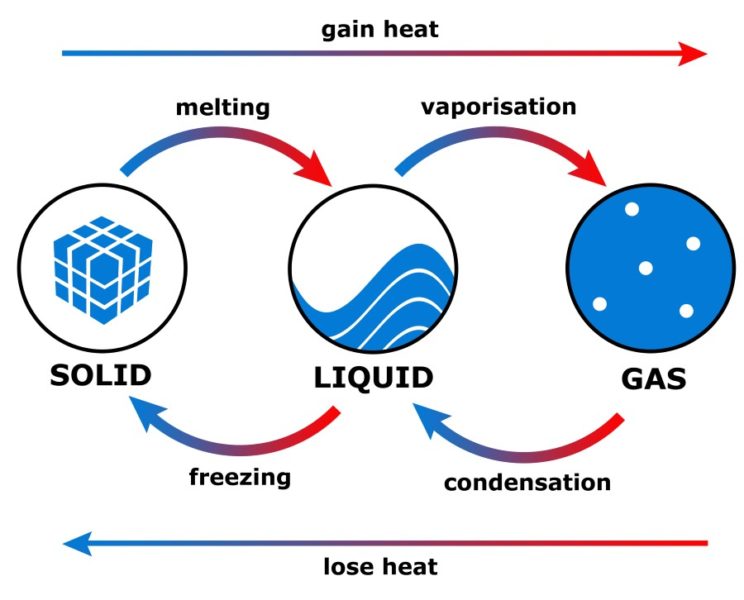
The second law of thermodynamics states that entropy always increases over time. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
